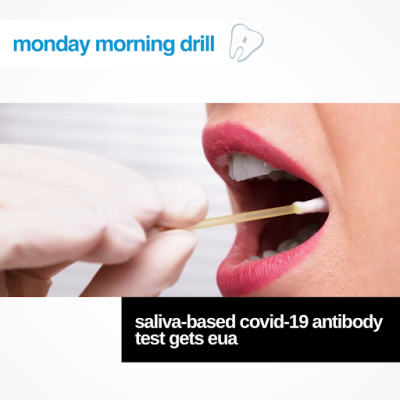
The FDA has given emergency use authorization to Diabetomics for its CovAb, a saliva-based COVID-19 antibody test that can deliver results within 15 minutes, FierceBiotech reported. CovAB is the first antibody test authorized by the FDA that uses mouth swabs instead of blood samples for detecting antibodies.
•
Do you think your schools or practices will begin using this salivary based test to screen patients if you’re still requiring patients to be tested prior to treatment? Let us know below!
•
Click the link in below to learn more about this study and what the test involves!
•
Check out weekly news and much more for FREE at TheToothBank.org ! We want to help you stay informed and up to date with all things in dentistry. ✔️
SOURCE: https://www.fiercebiotech.com/medtech/fda-greenlights-its-first-saliva-based-covid-19-antibody-test